*The picture in the header was taken from here.
This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison.
______________________________________________________________________________________________________
Small Molecule Interactions
Protein-protein interactions play a pivotal role in many biological processes [1]. Therefore, scientists have a keen interest to develop small molecules (as therapeutic antibodies) to regulate these interactions [1]. These small molecules have some advantages over antibodies because they are stable in human serum and they are specific to their target while antibodies do not penetrate cells easily and they have a high manufacturing cost [1]. While success in discovery and development of such molecules seem to promise a lucrative business, there are a few notable challenges such as the limited starting points of using small molecules to design drugs, the difficulty in differentiating real from artefactual binding, and the limited entries in typical small-molecule libraries [1].
PubChem was used to search for molecules that interact with the Ubiquitin-protein ligase E3A protein. Unfortunately, PubChem did not return any result. ChemBank was later used but it did not find any molecules interacting with our protein of interest.
A recent research by Huang et al. shows that there are 12 topoisomerase I inhibitors and 4 topoisomerase II inhibitors that can unsilence the paternal UBE3A allele. The list of the inhibitors can be accessed here. Since only the maternal allele is expressed in neurons, the unsilencing of the paternal allele could be a potential treatment for Angelman Syndrome patients, by compensating for the missing functional UBE3A gene. According to Huang et al., the paternal allele of UBE3A is silenced by a large antisense transcript (Ube3a–ATS). Due to allele-specific methylation of an imprinting centre that overlaps the Ube3a–ATS and Snurf/Snrpn transcription start site, this regulatory antisense RNA is expressed exclusively from the paternal allele. Huang et al. found that in cultured neurons treated with topotecan, there was an upregulated expression of UBE3A and a downregulated expression of Ube3a–ATS and Snurf/Snrpn. They also found that methylation at the imprinting centre remained the same. Therefore, it is proposed that topotecan unsilences paternal UBE3A by reducing the transcription of Ube3a–ATS, instead of altering the methylation state at the imprinting center.
Huang et al. also discovered that topotecan is able to unsilence UBE3A in neurons in vivo. Furthermore, the amount of protein expressed from the paternal allele is comparable to that expressed by the maternal control. Moreover, the effects of the drug treatment were still there even after 12 weeks. This shows that topotecan could potentially treat AS patients, besides treating cancer patients.
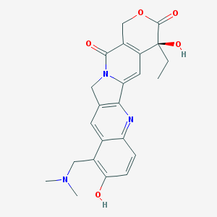
Figure 1: The 2D structure of topotecan. Diagram taken from PubChem.
References:
[1] Arkin, M. R., Wells, J. A. (2004). Small-molecule inhibitors of protein–protein interactions: progressing towards the dream. Nature Reviews Drug Discovery, 3: 301-317. doi:10.1038/nrd1343
[2] Huang, H., Allen, J. A., Mabb, A. M., King, I. F., Miriyala, J., Taylor-Blake, B., Sciaky, N., Dutton, J. W., Lee, H., Chen, X., Jin, J., Bridges, A. S., Zylka, M. J., Roth, B. L., Philpot, B. D. (2012). Topoisomerase inhibitors unsilence the dormantallele of Ube3a in neurons. Nature, 481: 185–189. doi:10.1038/nature10726
[3] PubChem
[4] ChemBank
If you find my website helpful, please consider donating to the Foundation for Angelman Syndrome Therapeutics (FAST)

Created by Jonathan Mok
[email protected]
Last updated 02/23/2012
Genetics 677

Created by Jonathan Mok
[email protected]
Last updated 02/23/2012
Genetics 677
